Sunbather:GMP 級 LNP 生產解決方案
將「Lipid Nanoparticles」(LNPs) 與其他奈米藥物推進至「Good Manufacturing Practice」(GMP) 階段,往往伴隨著高昂的成本與繁瑣的製程重新驗證。設備更換、流路變更與混合器差異,都可能導致批次品質不一致,延誤藥物上市時程。
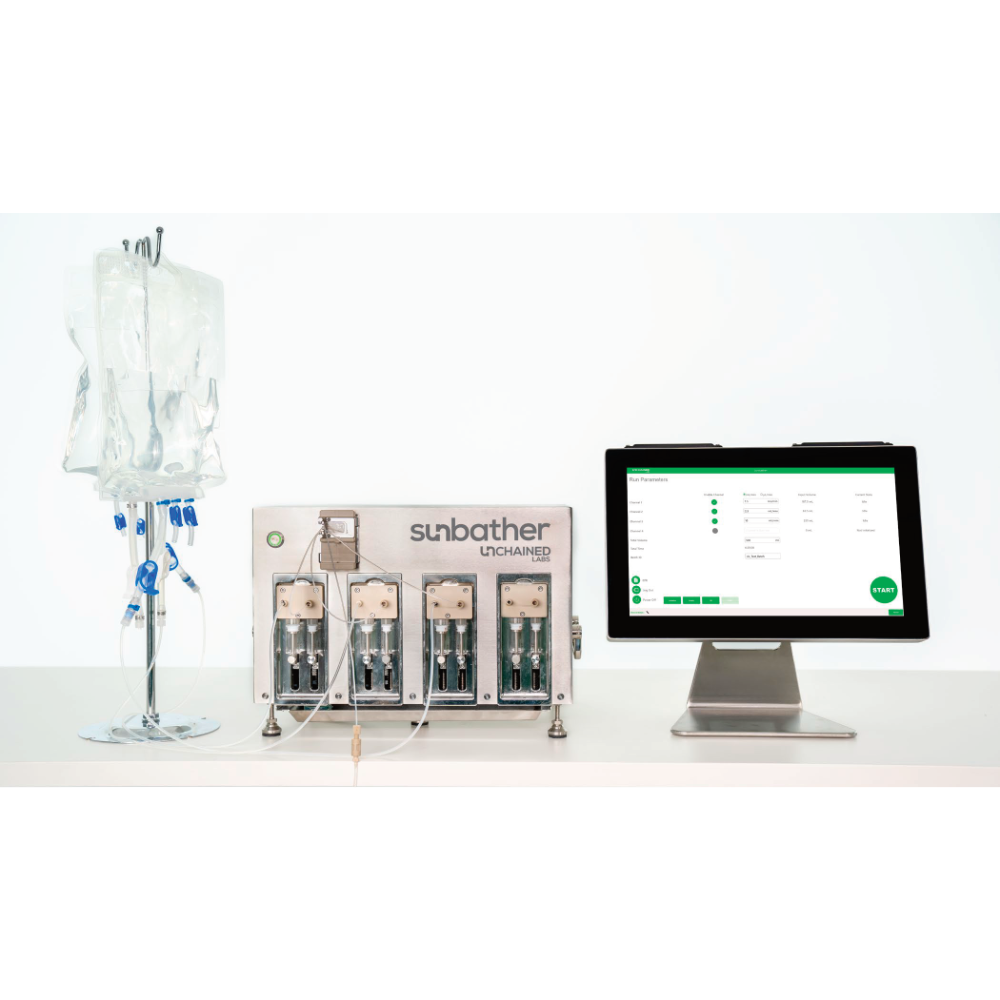
Sunbather 是 Unchained Labs 專為臨床製造設計的 GMP 級 LNP 生產系統。有別於傳統製程放大方案,Sunbather 採用與 Sunscreen 及 Sunshine 完全相同的泵浦技術與 Sunny 微流道晶片,確保研發階段鎖定的製程參數可直接轉用,無需額外優化或重新驗證。
從配方篩選到臨床生產,Sunny Suite 以一致的微流道混合技術貫穿整個開發流程,大幅降低製程轉移的失敗風險,節省時間與成本。

四大臨床級優勢
製程轉移零落差
Sunbather 使用與 Sunshine 相同的高精度連續式注射泵與 Sunny 微流道晶片。在開發階段確立的「Total Flow Rate」(TFR) 與「Flow Rate Ratio」(FRR) 等關鍵參數,可直接套用至 GMP 生產,無需重新優化製程。
GMP 法規合規設計
軟體完全符合「FDA 21 CFR Part 11」規範,具備完整的「Audit Trail」審計追蹤功能與分級權限管理(Administrator / Operator)。系統提供「IQ/OQ」驗證服務,並以品質管理系統(QMS)作為製程管控基礎。
污染防護與流路彈性
配備可完全更換的液體接觸流路(Replaceable Flowpath),包含泵浦閥、注射器與管路,並隨附「Extractables」數據供法規審查。支援單次使用(Single-use)或原位清洗(Clean-in-place, CIP)兩種模式。
高效能產出
單台產能可達每小時 1.8 公升。高效能連續式注射泵可長時間穩定運作,已驗證可生產公升級材料。若需更高產量,可透過多機並行方式擴充,無需變更製程條件。
Sunbather 核心效益
精準微流道控制
微流道混合技術在整個開發與製造過程中提供卓越的製程控制能力
泵浦技術一致
相同的高精度泵浦技術貫穿所有 Sunny Suite 儀器,消除製程轉移障礙
降低失敗風險
高可靠性製程減少批次浪費,節省時間與成本
完整法規文件
提供所有必要的追溯文件,確保符合法規要求
優質服務支援
從安裝培訓到售後技術支援,提供完整服務
加速上市時程
整合以上優勢,讓您的疫苗與治療藥物更快進入市場
Sunnies 微流道晶片:精密混合的核心
Sunnies 微流道晶片不僅是簡單的混合通道。每一片晶片皆由化學惰性玻璃製成,通道表面粗糙度僅 ±5 nm,遠優於傳統射出成型晶片的 ±1-3 µm,有效避免材料沉積與堆積。
精心設計的混合幾何結構可實現徹底且可控的混合效果。多種混合幾何選擇讓您可依據特定產品與製程需求,選擇最適合的流速與混合條件。GMP 級 Sunnies 與 Sunscreen 及 Sunshine 使用的晶片具有相同的混合設計,確保開發階段確立的製程可完美銜接至 GMP 生產。

Sunny Trident
- Reverse-angle mixing(逆角度混合)
- In-line dilution(管線內稀釋)
Sunny X
- Cross-type mixing(交叉式混合)
- Multiple channel sizes(多種通道尺寸)
Sunny T
- T-mixing(T 型混合)
- Multiple channel sizes(多種通道尺寸)
簡潔合規的操作軟體
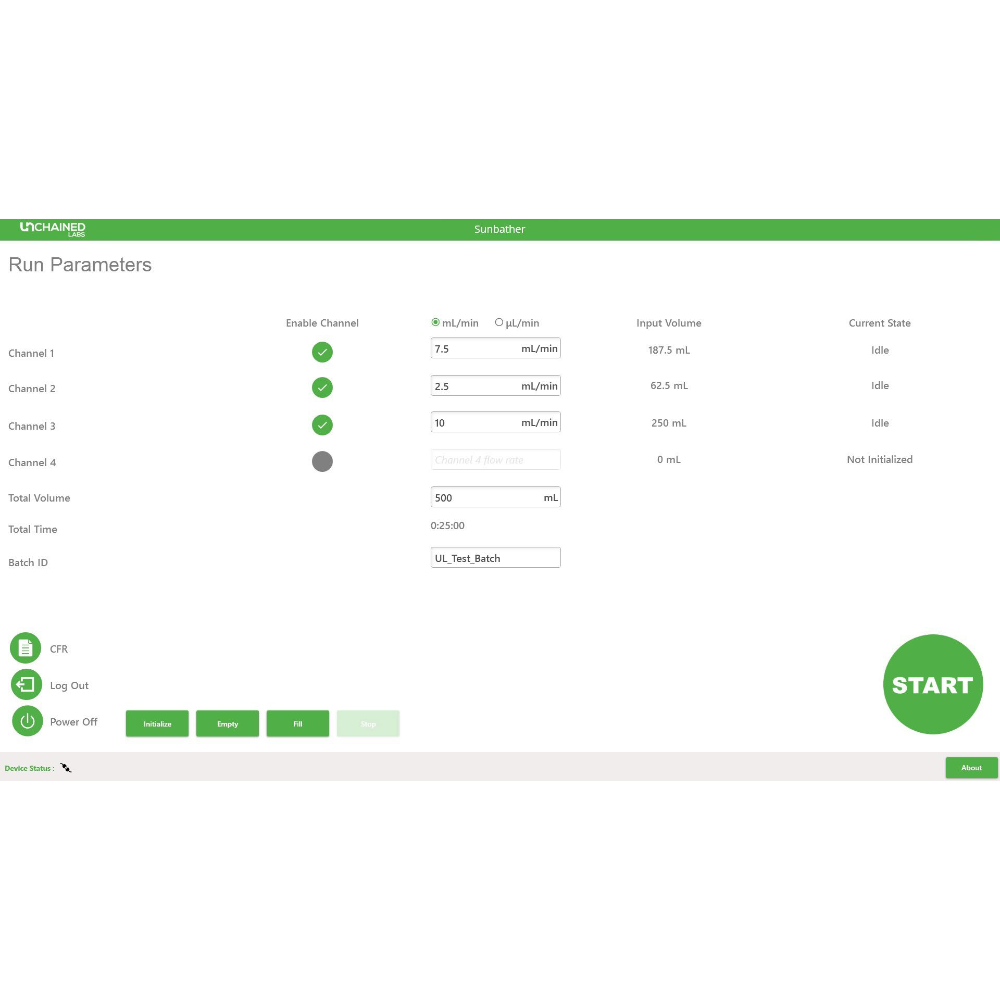
Sunbather 的軟體介面專為生產環境設計,提供 Administrator 與 Operator 兩種存取權限層級,並內建符合「FDA 21 CFR Part 11」規範的「Audit Trail」審計追蹤功能。所有製程關鍵操作皆需密碼簽核確認。
簡潔的使用者介面讓操作人員能快速上手,觸控螢幕式操作台設計適合潔淨室環境,讓設定新批次運行變得輕鬆便捷。
只需連接生物製程袋、輸入從 Sunshine 連續模式開發的流速參數,即可開始生產所需量的奈米粒子懸浮液,操作簡單無負擔。
Sunny Suite:完整的 LNP 開發解決方案
Sunny Suite 為奈米粒子開發的每個階段提供最佳解決方案。Sunscreen 可自動化進行高達 96 種配方的高通量篩選;Sunshine 則透過自動化實驗與連續流動模式,精確找出最佳混合製程條件。當您準備進入臨床試驗階段,Sunbather 將在完全符合 GMP 規範的環境中放大生產您的奈米粒子。

Screen 篩選
Sunscreen
高通量配方篩選,每日可處理多達 96 種配方
Optimize 優化
Sunshine
製程條件優化,支援 Protocol 與 Continuous 模式
Produce 生產
Sunbather
GMP 級臨床生產,每小時可達 1.8 公升
跨設備製程一致性驗證
透過相同的流體輸送系統與微流道混合技術,Sunny Suite 確保從配方篩選到製程開發再到 GMP 生產的每一次轉換都能平穩過渡。無論是篩選數百種配方或生產公升級臨床批次,Sunny Suite 都能提供完整的解決方案。
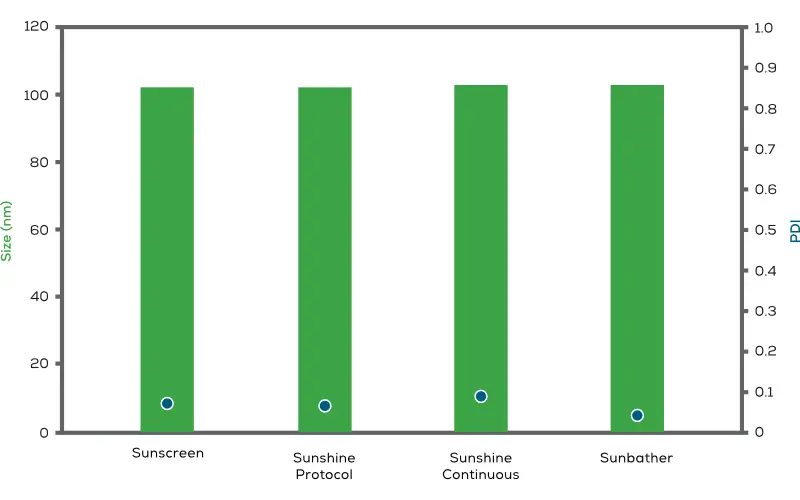
實驗數據顯示,使用相同配方與條件,在 Sunscreen、Sunshine(Protocol 模式)、Sunshine(Continuous 模式)及 Sunbather 上生產的 LNP 樣品,其粒徑(Size)、多分散性指數(PDI)與包封率(Encapsulation Efficiency, EE%)均維持高度一致性。
線上講座
Simplifying the transition from R&D to GMP-ready LNP production
「Lipid Nanoparticle」(LNP) 生產製程在從研發轉移至 GMP 環境時,經常面臨重大挑戰。設備更換導致資源浪費與繁重的製程重新驗證,為順利推進至下一階段帶來諸多阻礙。
本次線上講座將深入介紹 LNP 成功轉移至 GMP 生產的解決方案,包括符合 GMP 規範的觸控螢幕操作軟體,以及具備「Extractables」數據支援的完全可追溯、可更換液體接觸流路。
Sunbather 採用與 Sunshine(全方位 LNP 製程開發系統)相同的泵浦與混合技術,簡化了 GMP 轉移流程。透過確保一致性,無需進行製程轉移或重新優化,即可輕鬆過渡至每小時最高 1.8 公升的 GMP LNP 生產。
講座重點:
- LNP 簡單且一致地轉移至 GMP 生產的解決方案
- Sunbather 系統的關鍵功能與效益
- 從篩選到 GMP 的直接製程轉移
應用文獻
Send your LNP product development to lightspeed with the Sunny Suite
「Lipid Nanoparticles」正在改變醫學的面貌——從快速應對傳染病,到體內基因編輯與標靶蛋白質替代療法,LNPs 正引領醫療創新。儘管 LNPs 在臨床上已獲得驗證,但對藥物開發者而言仍存在挑戰,其中之一便是確保研發階段生產的材料與商業化生產的材料保持一致。
本應用文獻詳細說明 Sunny Suite 如何引導客戶完成藥物開發的整個流程——從篩選大量配方庫的第一步,一直到臨床材料的製造。透過實際案例展示相同配方在 Sunscreen、Sunshine 與 Sunbather 上生產時,粒徑、PDI 與包封率等關鍵品質屬性的一致性表現。

Technical Specifications
Application
| Throughput | 10 mL/min per channel |
| Total flow rate range | 0.1–30 mL/min (configuration dependent) |
| Flow rate ratio (aqueous to organic) range | 1:1 to 10:1 |
| Typical particle size range | 40–200 nm* |
| PDI | <0.2* |
| Encapsulation efficiency | >90%* |
Instrument - Physical
| Weight and dimensions (instrument only) | 19 kg, 29 cm H x 41 cm W x 32 cm D |
| Operating pressure | 0-10 bar |
Instrument - Electrical
| Voltage Input | 100 V–240 V AC, 50-60 Hz |
| Power consumption (peak/average) | 1A/500 mA |
Instrument - Other Information
| Fluidic port sizes | 1/4"-28 threaded nut, luer fitting (male and female) |
| Wetted materials | PEEK, FEP, Glass, PTFE, Silicone |
| Computer | Windows 11 Enterprise Touchscreen PC with Kiosk-mode operation |
Consumables
| GMP Sunnies | GMP-ready glass microfluidic mixing devices of various geometries and channel sizes |
| Available types | Sunny 490 Trident T, Sunny 100 X, Sunny 100 T, Sunny 190 X, Sunny 190 T, Sunny 275 X, Sunny 275 T, Sunny 150 3D, Sunny 50 Micromixer |
| Sunbather 1-Sunny Flowpath Pack | A complete wetted flowpath for use with the Sunbather instrument running a 1-Sunny configuration. |
| Sunbather 2-Sunny Flowpath Pack | A complete wetted flowpath for use with the Sunbather instrument running a 2-Sunny configuration. |
* Formulation dependent
常見問題
About Sunbather - GMP-Ready LNP Production System
Sunbather from Unchained Labs is the GMP-ready solution for lipid nanoparticle (LNP) clinical manufacturing. Designed to eliminate the costly and time-consuming process revalidation typically associated with scale-up to GMP production, Sunbather maintains exact process consistency with its Sunny Suite family members - Sunscreen for high-throughput formulation screening and Sunshine for process optimization.
The system features the same high-precision continuous syringe pump technology and Sunnies microfluidic mixing chips used throughout the Sunny Suite platform. This design philosophy ensures that process parameters developed during the R&D phase, including total flow rate (TFR) and flow rate ratio (FRR), can be directly transferred to GMP manufacturing without additional optimization or validation studies.
Sunbather delivers FDA 21 CFR Part 11 compliant software with comprehensive audit trail functionality and tiered access control (Administrator and Operator levels). The touchscreen kiosk interface is designed for cleanroom environments, enabling fast setup and intuitive operation with minimal training requirements.
Key features include a fully replaceable wetted flow path with accompanying extractables data, supporting both single-use and clean-in-place (CIP) protocols. The system offers throughput up to 1.8 liters per hour, with the capability for parallel operation when higher production volumes are required.
GMP-ready Sunnies are available in multiple geometries including Sunny Trident (reverse-angle mixing with in-line dilution), Sunny X (cross-type mixing), and Sunny T (T-mixing), each offered in various channel sizes to accommodate different flow rate requirements and applications. All chips are manufactured from chemically inert glass with ultra-smooth channel surfaces (±5 nm roughness) to prevent material buildup during extended production runs.
Sunbather is available with installation qualification and operational qualification (IQ/OQ) services, comprehensive user training, and full warranty and maintenance packages. The system is backed by Unchained Labs' Quality Management System (QMS), providing complete material traceability and documentation for regulatory compliance.
Applications include GMP production of mRNA vaccines, mRNA therapeutics, gene therapy vectors, siRNA delivery systems, and other lipid nanoparticle-based drug products. Typical performance specifications include particle sizes of 40-200 nm, PDI below 0.2, and encapsulation efficiency exceeding 90% (formulation dependent).
The Sunny Suite platform represents a complete end-to-end solution for nanoparticle drug development, from initial formulation screening through process optimization to clinical manufacturing. By maintaining identical fluid delivery and microfluidic mixing technology across all three instruments, Sunny Suite enables seamless transitions between development stages, reducing batch failures, saving time and costs, and accelerating the path to market for life-changing vaccines and therapeutics.